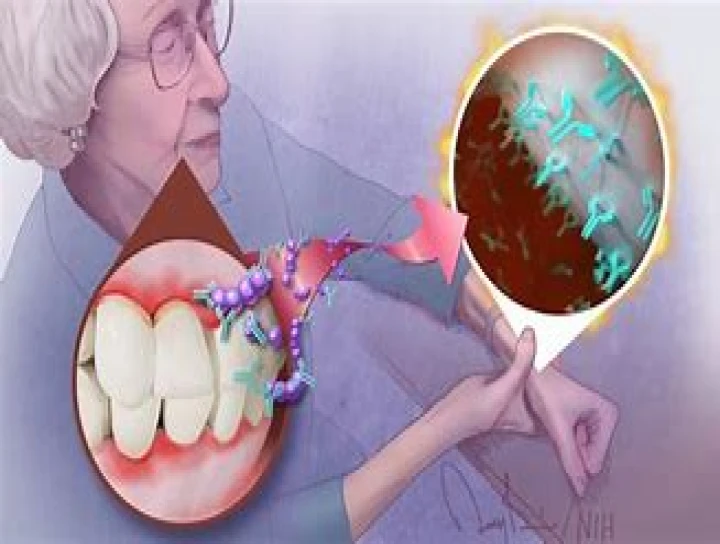
Chronic diseases often operate in isolation—or so it seems. But increasingly, research is revealing surprising connections between conditions once thought unrelated. One of the most compelling overlaps in recent years is the link between rheumatoid arthritis (RA) and periodontitis, a severe form of gum disease. Both are chronic, inflammatory diseases, and growing evidence suggests they share more than just similar symptoms: they may be driven by common immune system dysfunctions and inflammatory pathways.
Understanding how these two diseases intersect not only opens new avenues for treatment but also reinforces the importance of maintaining overall health—including oral health—to manage systemic conditions.
What is Rheumatoid Arthritis?
Rheumatoid arthritis is an autoimmune disorder that primarily affects joints. The immune system, which normally protects the body against infections, mistakenly attacks the synovium—the lining of the membranes that surround the joints. This leads to chronic inflammation, joint pain, swelling, and eventually joint damage and deformity.
RA is a systemic disease, meaning its effects go beyond the joints, impacting organs like the lungs, heart, and eyes. It is also associated with fatigue, anemia, and overall reduced quality of life.
What is Periodontitis?
Periodontitis is a chronic inflammatory disease of the gums and supporting structures of the teeth. It begins with the accumulation of bacterial plaque at the gumline. If left untreated, it can lead to gum recession, bone loss, and tooth loss.
While poor oral hygiene is a major factor, periodontitis is not just a local infection; it’s a disease of dysregulated inflammation. The body’s immune system responds excessively to the bacterial buildup, resulting in tissue destruction.
A Shared Inflammatory Mechanism
Both RA and periodontitis involve abnormal immune responses and chronic inflammation, and researchers are finding that these diseases may influence each other through several shared mechanisms:
1. Cytokine Overload
Cytokines are proteins that help regulate immune and inflammatory responses. In both RA and periodontitis, key pro-inflammatory cytokines—such as tumor necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), and interleukin-6 (IL-6)—are found in elevated levels.
These cytokines promote inflammation, tissue destruction, and bone resorption. For example, TNF-α not only contributes to joint damage in RA but also facilitates the breakdown of alveolar bone in periodontitis.
2. Neutrophil Dysfunction
Neutrophils are immune cells that form the first line of defense against infection. In both conditions, neutrophils are overactive and improperly regulated. They release enzymes and reactive oxygen species that damage healthy tissue while failing to clear pathogens effectively, perpetuating inflammation.
3. Autoimmunity and Molecular Mimicry
One of the more intriguing connections involves autoimmune responses triggered by oral bacteria. A particular bacterium, Porphyromonas gingivalis, which plays a central role in periodontitis, produces an enzyme called peptidylarginine deiminase (PAD). This enzyme can modify proteins through a process called citrullination.
In people with a genetic predisposition to RA, these citrullinated proteins are mistakenly targeted by the immune system, potentially triggering or worsening autoimmune activity. This process, called molecular mimicry, suggests a biological bridge between oral infections and autoimmune joint disease.
Clinical Evidence of the Connection
Numerous epidemiological studies have found that individuals with RA are more likely to suffer from periodontitis, and vice versa. A meta-analysis published in the Journal of Clinical Periodontology found that RA patients have significantly worse periodontal health compared to healthy individuals.
Moreover, treatment of periodontitis has been shown to reduce systemic inflammation and improve RA symptoms. In several studies, non-surgical periodontal therapy led to a decrease in disease activity scores and inflammatory markers like C-reactive protein (CRP) in RA patients.
Implications for Treatment
Recognizing the shared inflammatory pathways between RA and periodontitis changes how we approach both conditions:
- Integrated Care: Rheumatologists and dentists, particularly periodontists, should collaborate more closely. Periodontal evaluations should become routine for RA patients.
- Anti-inflammatory Therapy: Drugs like TNF inhibitors, used to treat RA, may also provide secondary benefits for gum health. Conversely, maintaining oral hygiene could reduce systemic inflammatory burden and possibly improve RA outcomes.
- Preventive Focus: Given the potential bidirectional relationship, preventing or treating periodontitis may reduce the risk or severity of RA, especially in genetically susceptible individuals.
The Bigger Picture: Oral-Systemic Health
The connection between rheumatoid arthritis and periodontitis is part of a larger shift in medical understanding—acknowledging that oral health is fundamentally linked to overall health. The mouth is not a separate entity but an integral part of the body, and chronic infections or inflammation in the oral cavity can have far-reaching effects.
To Conclude
Rheumatoid arthritis and periodontitis are two diseases that, on the surface, affect very different parts of the body. Yet, beneath that surface lies a shared inflammatory and immune-based pathway. From cytokine storms to molecular mimicry, these conditions speak the same biological language—one of chronic, misdirected inflammation.
For patients and healthcare providers alike, the message is clear: Treat the mouth to help the joints, and vice versa. As research continues to uncover the intricate connections between systemic and oral diseases, integrated care will become not just beneficial, but essential.


No Any Replies to “"Rheumatoid Arthritis and Periodontitis: A Shared Inflammatory Pathway"”
Leave a Reply